& Mechanism
Green Chem.
& Mechanism
Reaction & Reagents info
- Thionyl chloride (SOCl2) is commonly used to convert (a) acid to acid chloride and (b) alcohol to Chloride
- Other reagents that convert alcohols to chloride are oxalyl chloride, (COCl)2, PCl3 and PCl5
Advantages
- Inexpensive method on manufacturing scale
- The by-products liberated during the reaction are HCl and SO2, As both being gases, the reaction is usually clean
Disadvantages
- Similar to other sulphur compounds, handling SOCl2 is bit difficult, due to its peculiar unpleasant pungent smell.
- Handling of SOCl2 needs thorough safety studies before planning manufacturing scale
Useful Links on Reagent & Reaction:
- Deoxychlorination (Reagent Guide, ACS Green Chemistry Institute) – Green Chemistry information on various chlorinating reagents.
For review papers and other articles,
refer to the tab "References"
Mechanism
Mechanism

Additional details

General Procedure-1 (if alcohol is commercially available):
To a solution of carboxylic acid (1 eq.) in alcohol (20 Vol) at 0 oC is added thionyl chloride (2 – 3 eq.) and stirred at room temperature overnight. The reaction is monitored by TLC. After the completion of the reaction, excess thionyl chloride is distilled off and the residue is dissolved in DCM. The organic layer is then successively washed with water (10 Vol x 2), aq. NaHCO3 solution and brine solution, dried over sodium sulphate, filtered and concentrated under reduced pressure. The crude product is purified by column chromatography
General Procedure-2 (if alcohol is expensive or synthesized in-house):
To a solution of carboxylic acid (1 eq.) in DCM at 0 oC (10 Vol) is added alcohol (1 eq.), followed by dropwise addition of thionyl chloride (2 – 3 eq.) and stirred at room temperature overnight. The reaction is monitored by TLC. After the completion of the reaction, excess thionyl chloride is distilled off and the residue is dissolved in DCM. The organic layer is then successively washed with water (10 Vol x 2), aq. NaHCO3 solution and brine solution, dried over sodium sulphate, filtered and concentrated under reduced pressure. The crude product is purified by column chromatography
Note:
- Usually alcohol is used as a solvent. If it is methanol, it would result in methyl ester. Ethyl ester would be formed if the alcohol is ethanol
- If the alcohol can not be used in excess, DCM could be used as the solvent and alcohol as the reagent
- The by-products liberated during the reaction are HCl and SO2, As both being gases, the reaction is usually clean.
For more details on reactions and reagents,
refer to the tab "Reaction, Reagents and Mechanism"
Typical Procedure:
- Esterification of a carboxylic acid (ChemSpider) — Open access
- Acid to Ester-1 (OrgSyn) — Open access
For more details on large-scale reactions and OPRD procedures,
refer to the tab "Scale-up & Green Chem"
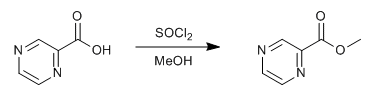
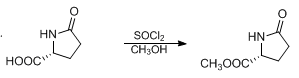
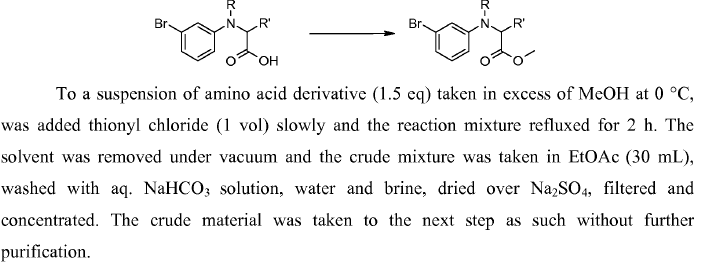
WO2010038081, page No. 203

WO2010038081, page No. 152
Green Chem.
- Synthesis of esters from carboxylic acids via 2 steps (acid chloride followed by alcohol) is one of common methods adopted for manufacturing
- Thionyl chloride could be carried out on large-scale. However, The by-products liberated during the reaction are HCl and SO2, Appropriate safety controls are to be ensured while performing manufacturing.
- Inexpensive method on manufacturing scale
Scale-Up Typical Procedure:
Green Chemistry Aspects:
- Deoxychlorination (Reagent Guide, ACS Green Chemistry Institute) – Green Chemistry information on various chlorinating reagents.