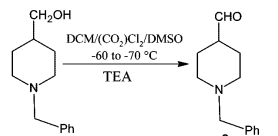
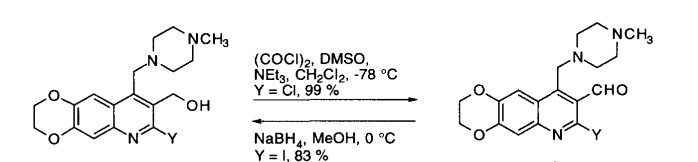
& Mechanism
Green Chem.
& Mechanism
Reaction & Reagents info
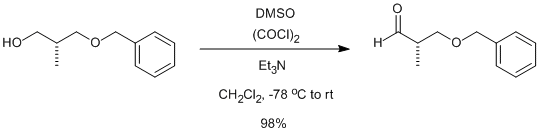
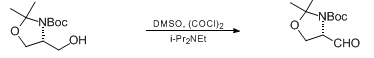
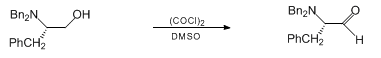
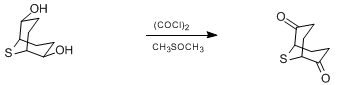
- Swern Oxidation (DMSO, Oxalyl chloride) is a mild oxidizing system that converts primary alcohol to aldehyde and secondary alcohol to ketone
- It is one of the series of oxidations involving DMSO (refer to DMSO-activated oxidations)
Advantages
- Inexpensive oxidation method on manufacturing scale
- Also, it is less toxic, compared to chromium-based alternatives (PCC and PDC)
Disadvantages
- The liberation of gases viz., malodrous dimethylsulphide (Me2S) and poisonous carbon monoxide (CO) are to be handled appropriately
- It is important to maintain the reaction mixture at -78 oC. If the temperature is not maintained, there is a possibility of formation of mixed thioacetals (see mechanism)
Useful Links on Reagent & Reaction:
- DMSO –Oxalyl Chloride (Swern Oxidation) (Reagent Guide, ACS Green Chemistry Institute) – Green Chemistry info.
- Swern oxidation (SynArchive) – Excellent compilation of reaction schemes with references
For review papers and other articles,
refer to the tab "References"
Mechanism
General Oxidation Mechanism
DMSO, as such, is not a good oxidising agent. However, it needs to be activated by an activator [such as (COCl)2 in Swern Oxidation, DCC in Moffatt oxidation] so that DMSO shall act effective oxidising agent (refer to DMSO-activated Oxidations)
Activation of DMSO by Oxalyl Chloride
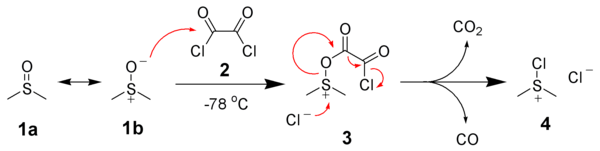
Swern Oxidation

Formation of side product in Swern Oxidation
In Swern oxidation employing (COCl)2, one mole each of CO, CO2 and Me2S (all gaseous products) are released. HCl gets converted to NEt3.HCl during the reaction
It is important to maintain the reaction mixture at -78 oC. If the temperature is not maintained, there is a possibility of formation of mixed thioacetals

Image from “chemistryworld.com”
Additional details

General Procedure:
To a solution of oxalyl chloride (2 eq.) in CH2Cl2 (10 Vol) at –78o C, DMSO (4 eq.) is added dropwise and the mixture iss stirred for one hour. Then, a solution of alcohol (1 eq.) in CH2Cl2 (5 Vol) is added at –78o C and stirred for another two hours. The reaction is monitored by TLC.
The RM is quenched by adding triethyl amine (8 eq.) at –78o C, followed by addition of water (10 Vol) at –78o C and stirred for 2 h. The reaction mixture is brought to RT and the layers are separated. The resultant organic layer is washed brine solution (5 Vol), dried over Na2SO4 and concentrated to afford the desired compound
Note:
- The most preferable solvent is DCM. THF can also be used
- It is usually performed at low temp (-78o C), and then stirred at RT
- The reaction mixture shall be warmed to -40 °C from -78o C, for secondary alcohols
- Bulkier amines such as DIPEA shall be used in place of triethylamine, if there is an epimerization of the α-center
- DMSO reacts violently with oxalyl chloride in the absence of a solvent and it can lead to an explosion.
For more details on reactions and reagents,
refer to the tab "Reaction, Reagents and Mechanism"
Typical Procedure:
- Swern Oxidation of an Alcohol (ChemSpider) — Open access
- Swern Oxidation-1 (OrgSyn) — Open access
- Swern Oxidation-2 (OrgSyn) — Open access
- Swern Oxidation-3 (OrgSyn) — Open access
For more details on large-scale reactions and OPRD procedures,
refer to the tab "Scale-up & Green Chem"
WO2016011390, page no. 112

Green Chem.
Swern oxidation could be carried out on large-scale. However, the reaction involves the liberation of 1 eq. each of the gases such as Me2S (dimethylsulphide), CO (carbon monoxide), CO2. Appropriate safety controls are to be ensured while performing manufacturing. During work-up, HCl gets converted to amine salt (such as NEt3.HCl).
- Swern Oxidation is one of the inexpensive methods to manufacture aldehydes or ketones from Alcohols
- The liberation of gases viz., malodrous dimethylsulphide (Me2S), poisonous carbon monoxide (CO) and CO2 are to be handled appropriately
- It is important to maintain the reaction mixture at -78 oC. If the temperature is not maintained, there is a possibility of formation of mixed thioacetals (see mechanism in General Info section)
Scale-Up Typical Procedure:
- An Improved and Efficient Process for the Production of Donepezil Hydrochloride (OPRD, 2008) – 4 Kg batch; 3.5 Kg of DMSO & 3.7 Kg of (COCl)2 have been used
- Convergent Catalytic Asymmetric Synthesis of Camptothecin Analog GI147211C (Tetrahedron, 1997) – 100 g batch
Green Chemistry Aspects:
Process Safety Aspects:
Swern Oxidation – References:
- The Swern Oxidation: Development of a High-Temperature Semicontinuous Process. Org. Process Res. Dev. 2008, 12 (5), 940-945.
- Convergent Catalytic Asymmetric Synthesis of Camptothecin Analog GI147211C. Tetrahedron 1997, 53, 10953
Activated DMSO Oxidation – Review: